The Microbiome Gambit
How EVG7 Rewrites the Nash Equilibrium of Antibiotic Resistance
The Strategic Failure of Broad-Spectrum Paradigms
As we navigate the first quarter of 2026, the global medical apparatus stands at a precarious juncture in its ongoing biological arms race against antimicrobial resistance (AMR). For nearly eight decades, the foundational doctrine of infectious disease management has relied upon a principle of scorched-earth pharmacology. Clinicians, operating under extreme time constraints, profound information asymmetry, and life-or-death stakes, have historically defaulted to the deployment of broad-spectrum antimicrobial agents. In the rigorous analytical framework of Game Theory, this dynamic represents a classic, deeply entrenched, and highly suboptimal Nash Equilibrium. A Nash Equilibrium describes a state within a strategic game where no individual player can unilaterally improve their own payoff by changing their strategy, provided the strategies of all other players remain constant. In the context of critical care medicine, no single physician is currently incentivized to deviate from prescribing the most aggressive, broad-spectrum agent available. The immediate payoff—rapid stabilization of the patient and avoidance of fatal sepsis—overwhelmingly outweighs the delayed, diffuse, and systemic cost of long-term antimicrobial resistance and collateral ecological destruction within the patient’s own body. However, while this strategy maximizes the immediate clinical payoff, it virtually guarantees a long-term failure state for both the individual host and the healthcare system at large. The human gastrointestinal tract, a vastly complex ecosystem comprising trillions of symbiotic microorganisms, is treated not as a cooperative biological asset, but as acceptable collateral damage.
This zero-sum approach to bacterial eradication has birthed a catastrophic secondary crisis: the proliferation of opportunistic pathogens that thrive in the biological vacuum left behind by broad-spectrum antibiotics. Chief among these is Clostridioides difficile (C. diff), a highly resilient, spore-forming bacterium responsible for debilitating and frequently fatal intestinal infections. When standard-of-care antibiotics such as vancomycin are deployed, they annihilate both the target pathogen and the commensal bacteria that normally hold C. diff in check. This strategy essentially burns down the forest to eliminate a single predator, leaving the sterile soil ripe for the predator’s eventual, undisputed return. The introduction of the experimental glycopeptide antibiotic EVG7 marks the historical pivot where precision biological architecture finally obsoletes the scorched-earth paradigm of broad-spectrum antimicrobials. Developed by Professor Nathaniel Martin’s research group at the Institute of Biology Leiden (IBL), EVG7 fundamentally breaks the existing Nash Equilibrium. By selectively targeting the pathogen while entirely sparing the critical, protective cohorts of the gut microbiome, EVG7 decouples immediate clinical efficacy from long-term ecological destruction. It offers a new Dominant Strategy—a course of action that results in a superior payoff regardless of what the opponent (the pathogen) chooses to do. In doing so, it rewrites the fundamental rules of engagement in modern microbiology.
The Macroeconomic Burden of Clostridioides difficile
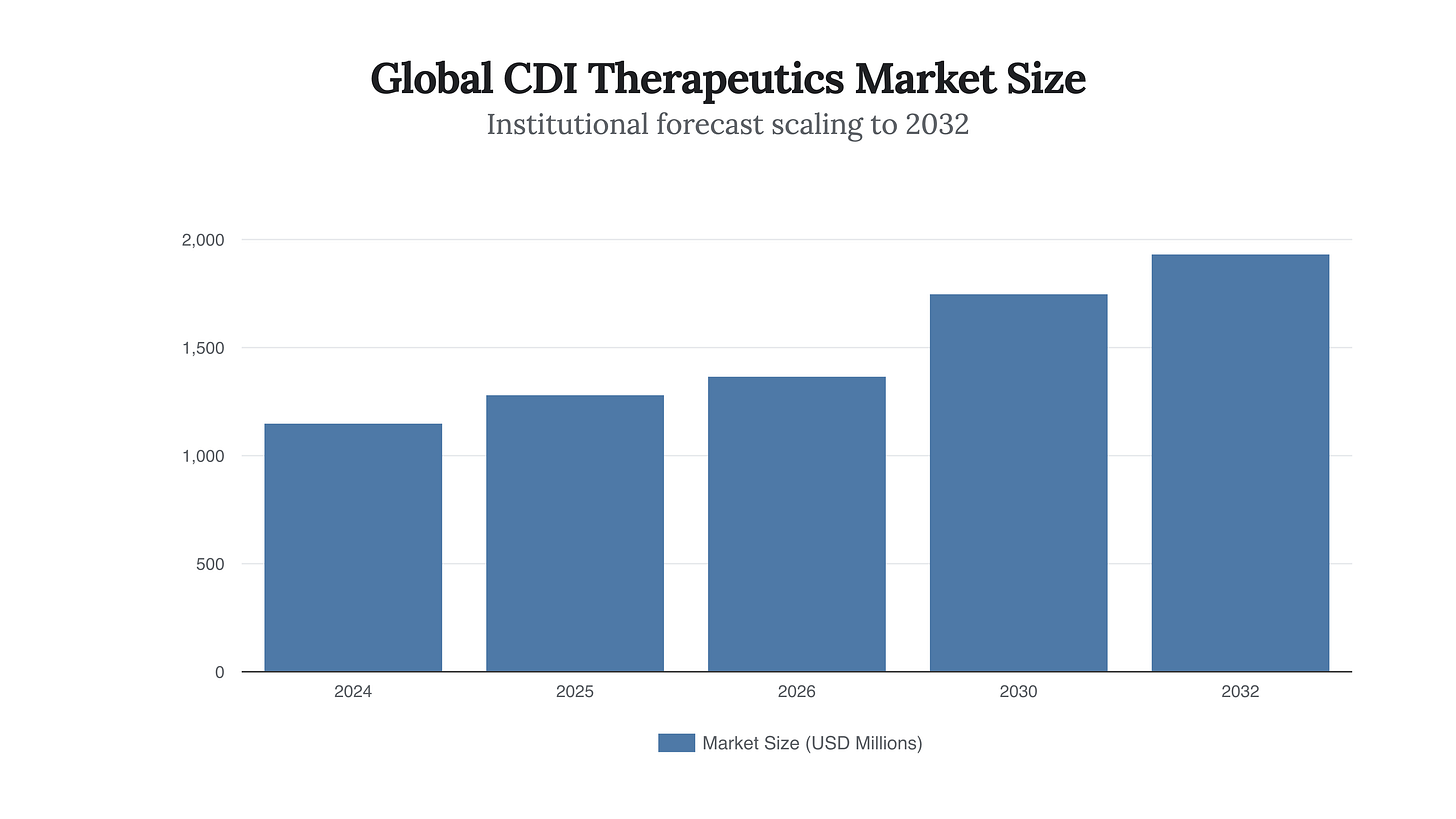
To understand the sheer magnitude of the breakthrough represented by EVG7, one must first deeply analyze the data reality of the Clostridioides difficile burden as it stands in March 2026. The economic and epidemiological toll of CDI is a compounding crisis that directly threatens the fiscal solvency of institutional healthcare providers. In 2025, the global market for Clostridium Difficile Infection therapeutics was valued at a staggering USD 1,285.43 million. Driven by the rising incidence of hospital-acquired infections and an aging demographic exceptionally vulnerable to gut dysbiosis, this market is projected to aggressively expand, reaching USD 1,934.09 million by 2032, advancing at a steady Compound Annual Growth Rate (CAGR) of 6.0%. Yet, these market size figures merely represent the surface-level expenditure on pharmaceuticals; they do not capture the devastating, secondary economic hemorrhage caused by the disease’s defining characteristic: its recurrence rate. The fundamental pathology of CDI makes it a uniquely expensive disease to manage. The bacterium produces powerful toxins (Toxin A and Toxin B) that trigger severe inflammation, profound diarrhea, and pseudomembranous colitis, frequently requiring extended intensive care admissions, strict isolation protocols, and highly specialized nursing care.
The true strategic failure of the current standard of care lies in the recurrence trap. Between 25% and 30% of all patients treated for an initial CDI episode with frontline therapies like vancomycin or fidaxomicin will experience a recurrent infection (rCDI) within weeks of completing their antibiotic course. With each subsequent relapse, the probability of yet another recurrence increases exponentially, creating a vicious, compounding loop of illness. In Game Theory, this scenario mirrors an Iterated Prisoner’s Dilemma played between the healthcare system and the pathogen. The hospital attempts to ‘cooperate’ with the patient’s recovery by deploying standard antibiotics. The pathogen, however, consistently ‘defects’ by retreating into dormant spores during the pharmacological assault, only to aggressively reactivate once the drug concentration falls below the minimum inhibitory threshold. Because the host’s indigenous microbiome has been obliterated by the treatment, the germinating C. diff spores face zero ecological competition, allowing them to rapidly reclaim dominance. Recurrent C. difficile is not merely a clinical failure; it is a compounding financial contagion that systemically drains hospital profitability under modern value-based care reimbursement models. Under these models, hospitals are heavily penalized for readmissions occurring within 30 days of discharge. A single episode of rCDI can easily consume upwards of $35,000 in unreimbursed critical care costs, annihilating the operating margins of the treating facility and transforming a treatable infection into a systemic financial liability.
Molecular Asymmetry: The Architecture of EVG7
To comprehend how EVG7 neutralizes the escalating threat of rCDI, one must examine the structural biology of the molecule through the lens of strategic asymmetry. EVG7 is not an entirely novel class of drug built from scratch; rather, it is a highly evolved, precision-engineered derivative of the well-established glycopeptide antibiotic vancomycin. Discovered in the 1950s in soil samples from Borneo, vancomycin operates by binding to the D-alanyl-D-alanine terminus of bacterial cell wall precursors, effectively halting cell wall synthesis and causing the bacterium to rupture. However, over decades of systemic overuse, its efficacy has plateaued, and its collateral damage profile has become unacceptable. Professor Nathaniel Martin’s team at Leiden University recognized that the base chassis of vancomycin possessed untapped potential. By executing sophisticated, targeted modifications to the molecular structure, they created EVG7—an experimental glycopeptide that binds with vastly superior affinity and selectivity. This molecular engineering introduces a profound Information Asymmetry into the biological battlefield. EVG7 possesses highly specific chemical ‘information’ that allows it to recognize and lock onto the cellular architecture of C. difficile with an unprecedented degree of precision, bypassing other bacterial structures entirely.
The quantitative validation of this molecular asymmetry is found in the Minimum Inhibitory Concentration (MIC) data published in Nature Communications in October 2025. In rigorous in vitro susceptibility assays, clinical isolates of C. difficile were proven to be up to 16 times more sensitive to EVG7 than to legacy vancomycin. Specifically, the MIC for EVG7 ranges from an astonishingly low 0.063 to 0.25 mg/L, compared to the 0.5 to 2 mg/L required for vancomycin. This exponential increase in potency constitutes a overwhelming Credible Threat in the strategic interactions between the host and the pathogen. A Credible Threat in Game Theory is a conditional strategy that is demonstrably enforceable; EVG7’s massive binding affinity guarantees that even trace concentrations of the drug will aggressively neutralize active C. diff vegetative cells. By achieving a 16-fold increase in binding affinity, EVG7 transforms vancomycin from a blunt, untargeted instrument into a molecularly guided biological munition. This hyper-potency is the critical prerequisite for the downstream strategic advantages of the drug. Because it is so intensely lethal to the target pathogen at microscopic concentrations, researchers were empowered to drastically alter the entire dosing paradigm, unlocking a secondary, immensely powerful ecological effect that standard antibiotics cannot safely achieve.
The Non-Zero-Sum Alliance of Lachnospiraceae
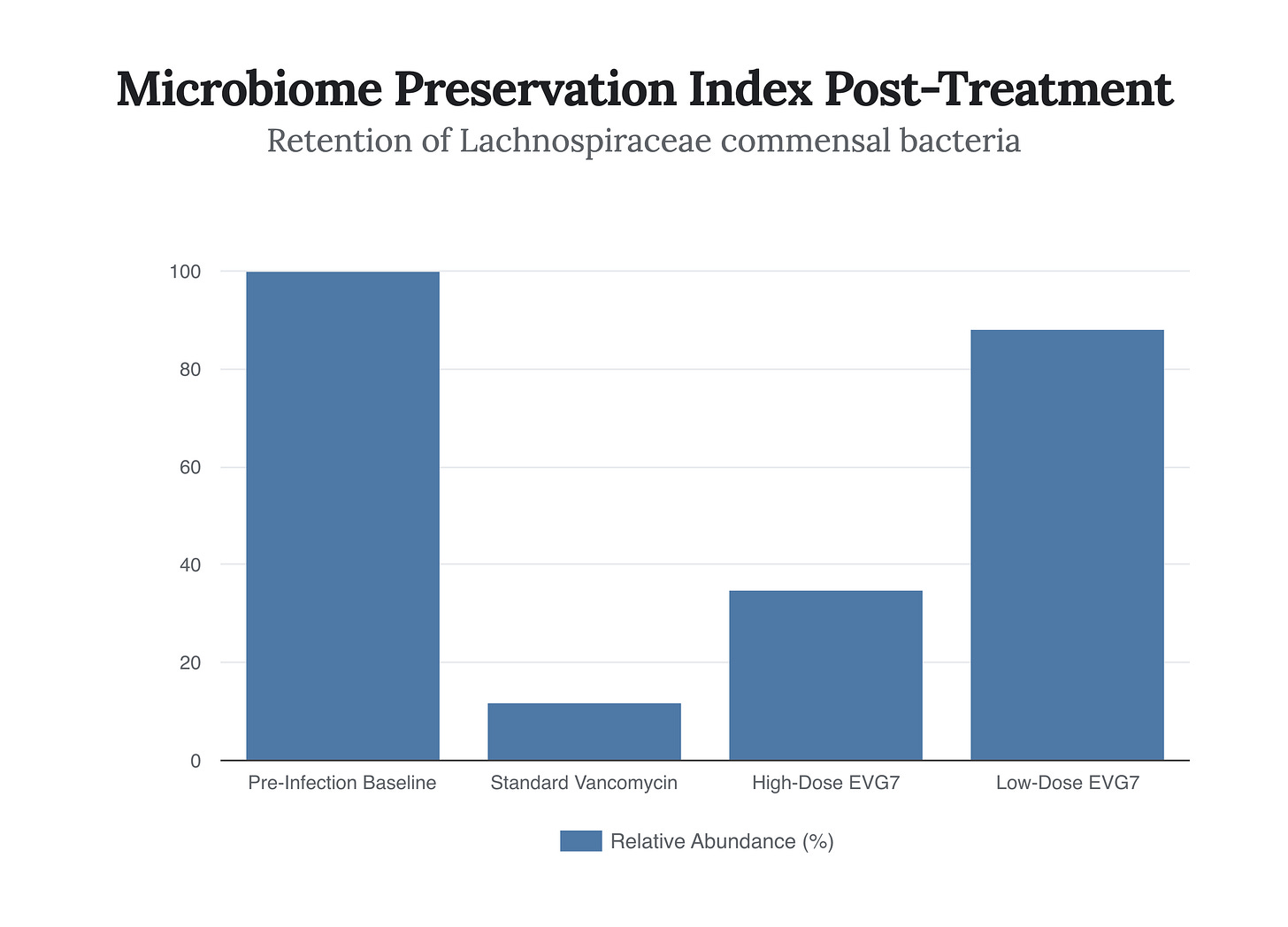
The true strategic brilliance of EVG7 extends far beyond its raw microbiological lethality; it resides in its capacity to alter the fundamental payoff matrix of the gut microbiome. In traditional antimicrobial therapy, the human gastrointestinal tract is treated as a Zero-Sum Game—a strictly adversarial environment where the complete eradication of all microbial life is deemed necessary to ensure the death of the specific pathogen. The host incurs massive collateral damage, losing the symbiotic bacteria that perform critical functions ranging from immune modulation to the synthesis of essential vitamins. EVG7 radically shifts this dynamic from a Zero-Sum Game to a Non-Zero-Sum cooperative alliance. By operating at exceptionally low doses, EVG7 displays an extraordinary capacity to selectively spare specific, highly beneficial cohorts of the commensal microbiome. Lead author Elma Mons and her colleagues demonstrated through comprehensive microbiome sequencing that mice treated with EVG7 retained robust populations of the Lachnospiraceae family. This is a crucial strategic vector, as Lachnospiraceae are not merely passive bystanders; they are active, aggressive defenders of the intestinal ecology.
Members of the Lachnospiraceae family are key producers of short-chain fatty acids (SCFAs), most notably butyrate. Butyrate serves as the primary energy source for colonocytes (the cells lining the colon), maintaining the structural integrity of the gut barrier and suppressing systemic inflammation. More importantly, the presence of these commensal bacteria creates intense competition for physical adherence sites and vital nutrients within the gut lumen, a phenomenon known as colonization resistance. When EVG7 selectively spares Lachnospiraceae, it effectively conscripts a biological army to guard the territory immediately after the active C. diff cells are neutralized. The Game Theory payoff matrix for the host is fundamentally transformed. Instead of emerging from treatment as a barren, defenseless landscape highly susceptible to reinvasion, the host emerges with an intact, aggressively territorial indigenous flora. EVG7 weaponizes the patient’s own microbiome, turning a passive biological ecosystem into an active, Non-Zero-Sum defensive perimeter against pathogen resurgence. This alliance ensures that the immediate clinical victory over the active infection is translated into permanent, long-term stability, dramatically altering the epidemiological trajectory of the disease.
The Dose-Response Paradox and Strategic Dominance
Perhaps the most intellectually provocative and counter-intuitive revelation from the Leiden research is the discovery of the Dose-Response Paradox. In the conventional pharmacological mindset, efficacy is generally assumed to correlate linearly with dosage, up to the point of toxicological limits. More drug equals more bacterial death, which presumably equals better clinical outcomes. However, the in vivo murine models utilized by Mons and her team shattered this assumption. When testing EVG7, they discovered that lower doses of the drug were vastly more effective at preventing recurrence than higher doses. In a validated mouse model of rCDI, an exceedingly low oral dose of EVG7 (0.04 mg/mL administered in drinking water) comprehensively outperformed both a 10-fold higher dose of legacy vancomycin and, crucially, higher concentrations of EVG7 itself. To analyze this through Game Theory, we must look at the concept of the Dominant Strategy. A Dominant Strategy is one that yields the highest payoff regardless of the variables introduced. In this biological game, prescribing a high dose of EVG7 becomes a self-defeating strategy. While it successfully eradicates the C. diff, the excess concentration crosses the selective threshold, inflicting collateral damage on the protective Lachnospiraceae.
By destroying the allied microbiome, a high dose removes the host’s secondary defense mechanism, rendering the gut vulnerable to eventual spore germination. The low dose, conversely, perfectly threads the pharmacological needle. Because EVG7 is inherently 16 times more potent than vancomycin, the low dose is still overwhelmingly lethal to the vegetative C. diff cells. Yet, it remains below the concentration required to harm the commensal flora. This creates a state of perfect Information Asymmetry; the drug provides just enough targeted chemical ‘information’ to collapse the pathogen without triggering a systemic ecological collapse. In the strategic calculus of EVG7, less is not more; less is the absolute Dominant Strategy that preserves the biological alliance required for permanent pathogen eradication. This biphasic, paradoxical response curve demands a fundamental re-education of clinical prescribing habits. Clinicians must abandon the heuristic that ‘hitting it hard’ is the safest approach for severe infections. The data reality dictates that surgical precision, executed through minimal effective dosing, generates vastly superior longitudinal outcomes. This requires institutional trust in the drug’s hyper-potency and a paradigm shift toward precision ecological engineering over brute-force chemical warfare.
Tit-for-Tat Microbiology and Spore Dormancy
To fully appreciate the strategic checkmate achieved by EVG7, one must deeply analyze the survival mechanics of Clostridioides difficile. The pathogen’s primary evolutionary defense mechanism is sporulation. When the bacterial colony senses extreme environmental stress—such as the introduction of a toxic glycopeptide antibiotic—the vegetative cells rapidly undergo a complex metamorphosis, condensing their genetic material into highly resilient, metabolically dormant spores. These spores are virtually impervious to standard chemical assaults, heat, and radiation. In the framework of Game Theory, sporulation is a classic execution of a Tit-for-Tat strategy over repeated interactions. When the clinician attacks (Defects), the bacterium retreats into its armored spore state (Defects), ensuring mutual stalemate. The clinician assumes the infection is cleared because the active symptoms subside and vegetative cells disappear from assays. However, as soon as the antibiotic is metabolized and flushed from the gastrointestinal system, the spore senses the return of a favorable environment. It immediately germinates (Cooperates with its own biological imperative), rapidly repopulating the now-sterile gut and triggering a devastating recurrent infection.
Traditional antibiotics fail because they cannot break this Tit-for-Tat loop; they only temporarily suppress the active phase while simultaneously preparing the perfect, competition-free environment for the spore’s eventual return. EVG7 dismantles this strategy entirely by introducing a third variable into the matrix: the retained commensal microbiome. While EVG7, like vancomycin, cannot directly kill the dormant spores, its low-dose precision leaves the Lachnospiraceae vanguard fully intact. When the EVG7 course concludes and the surviving C. diff spores attempt their inevitable germination sequence, they do not wake up in a sterile, undefended landscape. Instead, they find themselves immediately surrounded by a dense, aggressive, and established commensal population that physically blocks attachment sites and consumes all available bile acids and nutrients required for pathogen replication. The true genius of EVG7 lies not in killing the active bacterium, but in permanently checkmating the dormant spore’s Tit-for-Tat strategy through competitive ecological exclusion. The spores are effectively trapped in a hostile environment where germination equals immediate starvation and death. The credible threat of the spore is entirely neutralized, breaking the cycle of recurrence and shifting the ultimate payoff permanently in favor of the host.
Unlock deeper strategic alpha with a 10% discount on the annual plan.
Support the data-driven foresight required to navigate an era of radical uncertainty and join a community of institutional-grade analysts committed to the truth.